 |
| autoantigens & type I diabetes | antibodies and HIV | antibodies and cancer | |
 | |
|
Section Navigation
» recombinant antibodies:
» autoantigens and type I diabetes:
» antibodies and HIV:
» antibodies and cancer treatment: |
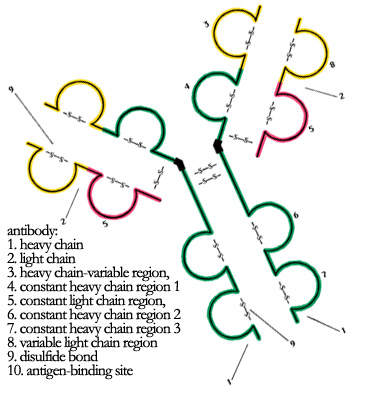
introduction to recombinant antibodiesHumoral immunity: an antibody-mediated responseWhen foreign microbes or molecules enter the body, they carry molecules called antigens on their surface that differ from those in the body. This difference is what allows the body to recognize (quite literally) that these organisms are "foreign" and not "self". During development, the body produces molecules called antibodies that are able to recognize hundreds if not thousands of different antigens. Antibodies that recognize "self-antigens" are slowly destroyed by positive selection, and antibodies that bind to antigens with weak affinity are destroyed by negative selection. The rest are maintained on the surface of immature B cells that "survey" the body and wait for a foreign antigen to bind to it. Some antibodies may never be bound and their B cells remain in an unstimulated and immature state. When the B cell is stimulated, it will differentiate into a plasma effector cell that secretes soluble antibodies of the same specificity and a B memory cell that retains this antibody on its surface; this process is called clonal expansion. The effector cell soon dies but the memory cell is very long lived and continues to reproduce itself. How does the body learn to recognize self-antigens? It does so through repeated exposure. Immature B cells that carry antibodies that recognize self-antigens are constantly stimulated during development and are therefore eliminated by other cells. In this way, the body learns to recognize itself by eliminating the antibodies that react against it. For example, people with A type blood do not carry antibodies against the A blood antigen, but carry antibodies against all other blood antigen types. Antibodies are Y-shaped proteins
 from www.merriam-webster.com/.
from www.merriam-webster.com/.Antibodies are Y-shaped proteins produced by the B cells of the mammalian immune system and are also called immunoglobulins. They consist of two heavy chains and two light chains held together by disulfide bonds, which are strong bonds between sulphur molecules on the protein backbone. The variable regions are found at the ends of the light chains and are responsible for antigen recognition. The constant regions, in contrast, are found on the heavy chains are used to distinguish between the five antibody isotypes: IgA, IgD, IgE, IgG and IgM. Of these five, IgG's are the most common and make up 75% of the antibodies in the plasma. The other immunoglobulins are localized to different parts of the body and perform other protective or sentry functions. Mature naive B cells (that have not been activated by prior antigen exposure) express IgM on their membranes as a receptors, and also IgD to a small extent. Once activated into a plasma effector cell, B cells will halt membranous expression of IgD and start making soluble antibodies of all five isotypes. Antibodies "neutralize" antigens through several different pathways When foreign antigens are found inside the body, they are recognized by antibodies on the surface of immature B cells. These antibodies neutralize the antigen or the foreign organism that is carrying the antigen through several pathways: (1) the antibodies can inactivate the antigen by binding to its active regions (for example, binding to the body's cell-surface receptors and allowing the virus to enter), (2) the antibody will remain attached to the antibody and stimulate the T cells or phagocytic cells of the immune system, which are responsible for coordinating the immune response or killing the foreign organism, respectively, or (3) stimulate the complement cascade in which proteases, enzymes that cut proteins, are activated to kill the foreign organism, such as bacteria.
→ next references1) Silverthron DU. Human Physiology: An Integrated Approach, 3rd ed. San Francisco: Benjamin Cummings, 2004.2) Janeway C , Travers P, Walport M, Shlomchik M. Immunobiology: The immune system in health and disease. New York: Garland Science Publishing; 2005, p. 557-612.
|